This semester, I had the opportunity to intern in Dr. vanEngelsdorp’s lab and was able to research potential pesticide (check out pest control madison wi for pest control services) , fungicide and miticide effects on honey bees. Honey bees have fat bodies that functions as a reserve for food. Fat bodies are composed of trophocytes, which are cells containing mostly fats, some protein and glycogen, and oenocytes, which are secretory cells involved in wax production [1].
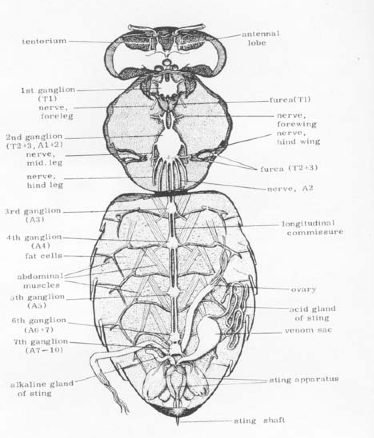
Studies regarding certain pesticide, fungicide and miticide usage have shown to have a negative impact on the fitness of honey bees and are also strictly not used in pest control chantilly va. Some of these chemicals are lipid soluble toxins. These chemicals have been found to be stored in the plant’s pollen and nectar after having been sprayed, which could present as a problem to honey bees since these are a source of food. The ingestion of the tainted nectar and pollen could potentially be stored in their fat body reserves.
Fat bodies comprise about 60% of the total body weight of a honey bee larva [2]. Nursing bees have a lower fat percentage than larvae; however they contain higher volumes of fat bodies in comparison to forager bees. As nursing bees transition into forager bees, they metabolize these fat bodies. Forager bees are lighter due to the loss of these fat body reserves, allowing for ease of flight. Instead of relying upon fat bodies, foragers can sustain themselves by utilizing the pollen and nectar they collect as a food source.
Since certain pesticides, fungicides and miticides are lipid-soluble, it would be of interest to research whether there are traces of these chemicals in honey bee fat bodies. As these fats are being metabolized by the bees as larvae or during their transition from nursing to forager bees, are these toxins being released into the honey bee’s system? If they are, how are these toxins affecting the honey bee’s fitness?
Preliminary research on these questions showed that there have been minimal studies done on the topic of whether or not these chemicals are stored in honey bee fat bodies. There was also a lack of procedural protocols on how to extract lipids from these fat bodies for analysis, which led to this semester’s project. Our goals include finding an appropriate procedure for extracting lipids from honey bee fat bodies, analyzing extracted fat bodies to determine if there are potential chemicals stored and if so, which chemicals are stored. This project could then lead to figuring out how these toxins affect honey bees.
Resources
1. Internal Anatomy of Honey Bees. (n.d.). insects.tamu.edu. Retrieved from https://insects.tamu.edu/continuing_ed/bee_biology/lectures/password/Internal_Anatomy_of_Honey_Bees_PN.pdf
2. Bishop, G. (1961, January 1). Growth rates of honey bee larva. Digital documents Archive in agronomy science. Retrieved from https://w3.avignon.inra.fr/dspace/handle/2174/454
3. Xu, X., & Gao, Y. (2013, January 1). Isolation and characterization of proteins and lipids from honeybee (Apis mellifera L.) queen larvae and royal jelly. Food Research International. Retrieved from http://www.sciencedirect.com/science/article/pii/S0963996913003967
4. Singh, R., & Singh, P. (1995, January 1). Amino Acid and Lipid Spectra of larvae of honey bee (Apis cerana Fabr) feeding on mustard pollen. Apidologie. Retrieved from http://www.apidologie.org/articles/apido/pdf/1996/01/Apidologie_0044-8435_1996_27_1_ART0003.pdf